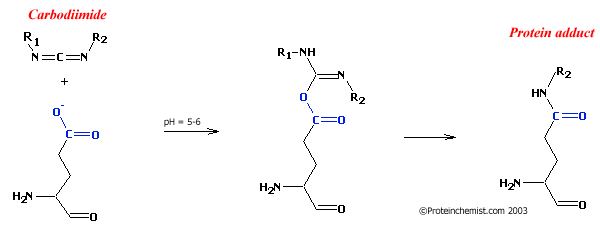
Figure 1: Reaction of carbodiimides with carboxylates
Carbodiimides are commonly used to modify proteins on their carboxylate groups. They react with proteins at about pH = 5 to 6 and can be used either alone or in combination with amines to create new amide bonds off the carboxyl. As seen below, isourea is formed in the first step of the reaction. This intermediate can either react with other amines in the solution or rearrange itself to form a stabile amide. (Step 2 in Figure 1, below) An exmple protocol for the modification of carboxylates would be to have the protein at pH=5.0, add 100 fold excess of a carbodiimide such as 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) and allow it to react overnight at 4 degrees.

Figure 1: Reaction of carbodiimides with carboxylates
In the presence of a diamine such as ethylenediamine the carboxyl can be changed to an amine functional group with a longer reach, a wider variety of commercially available modifying reagents, and a possible reversal of charge (See Figure 2).

Figure 2: Conversion of carboxylates to amines with carbodiimides